Press Release: DLRC Launches HTA Support to Accelerate Market Access
Published Aug 20, 2025
Published 29th July 2025

The appropriate determination of the condition for your paediatric application has important strategic implications. It can impact the scope and timelines of any paediatric development as well as the likelihood of gaining the reward, which is currently a six-month extension of the supplementary protection certificate, or for orphan medicines, an additional two years of market exclusivity, where a Paediatric Investigation Plan (PIP) is agreed and subsequently completed.
The Paediatric Regulation (Regulation (EC) No 1901/2006) frequently mentions the concept of the ‘paediatric condition’, yet it does not define the term. A legal definition of ‘condition’ is provided in the European Commission Guideline 2008/C 243/01 [2] as “any deviation(s) from the normal structure or function of the body, as manifested by a characteristic set of signs and symptoms (typically a recognised distinct disease or a syndrome)” ([2], section 1). However, this definition does not connect the concept with the intended adult indication, as is necessary in order to define a paediatric condition.
To address this gap, the Policy on the determination of the condition(s) for a Paediatric Investigation Plan/Waiver (Scope of the PIP/waiver) was developed to set out a systematic and consistent approach to determine the condition(s) of a PIP in relation to the proposed adult indication(s). In this blog, we discuss the paediatric condition, how it’s determined within the EU and our recent experience.
The paediatric condition refers to the therapeutic area in which the drug is likely to be of value in the paediatric population. It will include one or more potential paediatric indications and may or may not overlap with the intended adult indication. In effect, the Paediatric Committee (PDCO) can ‘go looking’ for further areas of potential paediatric development beyond the intended adult indication. This approach allows paediatric development to go beyond the often narrow scope of the proposed adult indication, addressing potential unmet needs and anticipated paediatric uses based on the product’s properties. On the other hand, a very broad paediatric condition would require extensive paediatric development, so in determining the paediatric condition, PDCO need to balance these two conflicting objectives against each other.
The PDCO defines the paediatric condition as the scope of paediatric development outlined in the PIP or waiver they agree upon. The policy also considers that PDCO may only request development in a single paediatric indication under a single paediatric condition. With that in mind, the definition of the paediatric condition and the agreements with PDCO that are established become particularly important when further adult development is planned.
For example, a PIP or waiver with a broad paediatric condition limits the requirement for further paediatric development should the Sponsor later pursue an additional adult indication which is considered to fall within the scope of the paediatric condition for which a PIP or waiver has already been agreed. In other circumstances, for example, when limited adult development is planned, it may be advantageous for a narrow paediatric condition to be agreed. This could mean that fewer studies are required to complete the PIP.
The following elements determine the paediatric condition:
It is the combination of all these aspects that determines the paediatric condition, which explains the complexity. This is where experienced regulatory professionals can guide clients through this process. The policy recommends using MedDRA as the chosen hierarchical system, with the condition generally identified at the High Level Term (HLT).
As a high-level overview, the MedDRA framework consists of five levels. The highest level, System Organ Class (SOC), is the broadest level and represents the body systems or organs affected, for example, ‘Respiratory, thoracic, and mediastinal disorders’. A High Level Group Term (HLGT) includes one or more HLT, creating a bridge between the SOC and HLT, for example, ‘Upper respiratory tract disorders.’ HLT describes one or more Preferred Terms (PT), for example, ‘Asthma and related conditions’. A PT is the level most commonly used for coding and data entry. It denotes either a symptom, sign, disease, diagnosis, therapeutic indication, investigation, surgical or medical procedure or medical, social or family history characteristic, for example, ‘Asthma’. The Lowest Level Term (LLT) is the most precise term, for example, ‘Bronchial asthma’.
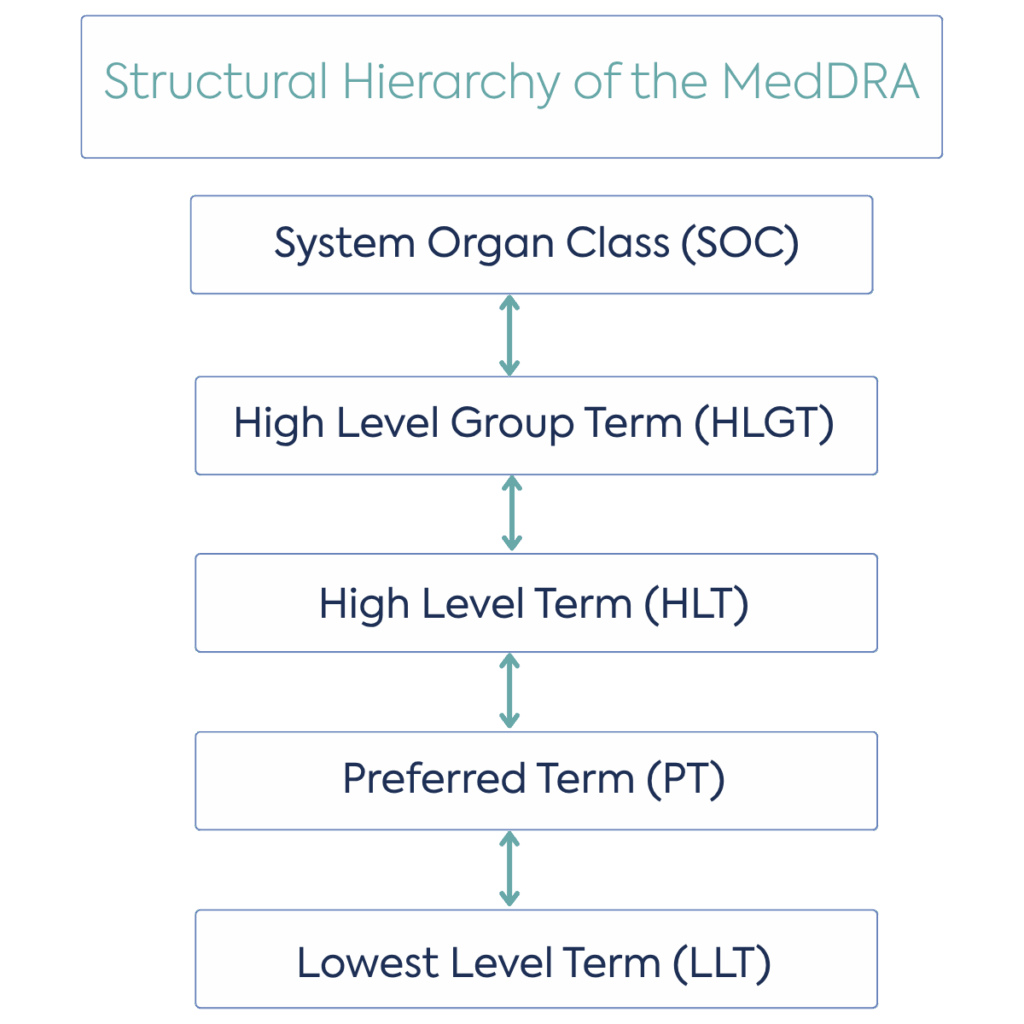
Figure 1. The structural hierarchy of the MedDRA, figure adapted from the Policy on the determination of the condition(s) for a Paediatric Investigation Plan/Waiver (scope of the PIP/waiver)
Considering the different levels in the MedDRA framework, it is the HLT which relates to the proposed indication in adults. Once determined, this HLT and all PTs falling beneath it, would be considered for potential paediatric use and, therefore the ‘paediatric condition’, also taking into account the other elements listed above.
Due to the varying granularity of MedDRA classification across different SOCs, PDCO can, in certain circumstances, request a higher (HGLT) or lower (PT) level to ascertain the paediatric condition, but would support the proposal with scientific justifications. Ultimately, defining the paediatric condition requires a nuanced understanding of both regulatory expectations and the MedDRA hierarchy, particularly the High Level Term, which anchors the scope of development.
To establish a hierarchical classification structure that supports the determination of the paediatric condition, PDCO evaluated different medical classification systems. These include the ICD (International Classification of Diseases) system and MESH (Medical Subject Headings) frameworks. Different hierarchical classification systems have been designed for different purposes, but none have been created with the objective of defining the paediatric condition.
Finally, it was recommended that MedDRA (the Medical Dictionary for Regulatory Activities) be used as a hierarchical classification structure to support the determination of the paediatric condition as it provides a consistent hierarchical structure across therapeutic areas and an appropriate level of granularity for determination of the paediatric condition. Furthermore, MedDRA is already used for certain regulatory activities.
At DLRC, we stay up to date with the latest regulatory intelligence in order to provide the best advice to our clients. Recently, we’ve observed that some paediatric applications for oncology products have referenced a WHO classification system, specifically WHO-HAEM5. WHO-HAEM5 is the 5th edition of the World Health Organization Classification of Haematolymphoid Tumours.
As described in a short review from the Magazine of European Medical Oncology, the latest WHO-HAEM5 classification includes updates primarily based on new molecular insights and places greater emphasis on the hierarchical structure. It’s possible that following consideration of these updates, PDCO will consider that although not foreseen in the policy, WHO-HAEM5 offers some utilisation over, or alongside MedDRA, in determining the paediatric condition at this point in time. However, it is still unclear whether this indicates a longer-term shift away from using MedDRA, at least for oncology conditions, and potentially toward a policy update.
In summary, defining the paediatric condition is a critical step in the development of a PIP/waiver application, with far-reaching implications for regulatory strategy, study design, and ultimately, the availability of medicines for children. While the MedDRA classification system remains the primary tool for this purpose, emerging trends – such as the use of WHO-HAEM5 in oncology – highlight the evolving nature of regulatory expectations. Sponsors must therefore remain agile, informed, and strategic in their approach. With expert guidance, such as that offered by DLRC, companies can navigate this complexity to ensure that paediatric development is both scientifically sound and aligned with regulatory requirements leading to fewer questions from health authorities and a more streamlined PIP/waiver procedure. Contact us today to discuss how we can support you in this evolving regulatory environment by emailing hello@dlrcgroup.com.

Published Aug 20, 2025

Published Jul 29, 2025

Published Jul 29, 2025

Published Jul 21, 2025

Published Jul 11, 2025

Published Jul 07, 2025

Published May 29, 2025

Published May 29, 2025

Published May 29, 2025