Using Out-of-Specification ATMP Batches: EMA’s 2026 Q&A
Published Mar 20, 2026
Published 26th February 2026

The introduction of the EU Health Technology Assessment Regulation (EU) 2021/2282 marks a fundamental shift in how clinical evidence is evaluated for market access across Europe. With the phased implementation of Joint Clinical Assessments (JCAs) beginning in January 2025 for oncology medicines and advanced therapy medicinal products, developers are entering a new, harmonised system for clinical assessment that runs in parallel with the European Medicines Agency’s marketing authorisation process.
While the Regulation aims to reduce duplication across Member States, it also introduces new strategic and operational demands. Evidence planning must now accommodate EU-level clinical assessment alongside national appraisal requirements, compressed timelines, and PICO-driven scoping that defines the precise questions developers must answer.
This article explores the strategic implications of the EU HTA Regulation for pharmaceutical companies preparing for European launch. Drawing on DLRC’s integrated regulatory and HTA expertise, we examine how JCAs reshape evidence planning, why early alignment between regulatory and access strategies is now essential and what practical steps sponsors can take to reduce risk, support reimbursement outcomes, and achieve timely, sustainable access across EU markets.
By the end of this article, readers will understand why the EU HTA framework represents more than a procedural update, how it affects clinical development and comparative evidence generation and how to position programmes for success under the new European market access landscape.
European market access has traditionally relied on national HTA processes, often resulting in duplicated clinical assessments and divergent evidence requirements across countries. The EU HTA Regulation introduces a single EU-level Joint Clinical Assessment intended to provide a common clinical evidence base for Member States, while leaving pricing, reimbursement and economic evaluation at the national level.
From January 2025, JCAs apply to new oncology medicines and ATMPs, expand to orphan medicines in 2028 and cover all centrally authorised medicines by 2030 (Figure 1). Running in parallel with the EMA centralised procedure, the JCA compresses end-of-development timelines and significantly raises the value of early, integrated evidence planning that is both regulatory robust and HTA-ready.
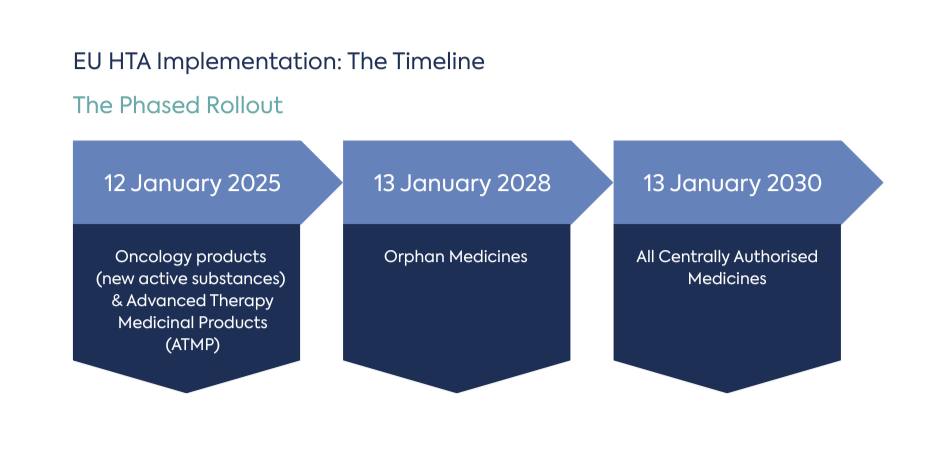
Figure 1. Timeline showing phased JCA implementation from 2025 to 2030 alongside EMA procedures.
A Joint Clinical Assessment is a coordinated EU-level evaluation of a health technology’s relative clinical effectiveness compared with relevant alternatives. It is explicitly clinical in scope. Economic evaluation, pricing decisions and final reimbursement remain the responsibility of individual Member States.
The JCA is closely anchored to the EMA timeline. Following validation of the marketing authorisation application, HTA bodies undertake PICO-based scoping to define the evidence questions the developer must address. Dossier preparation is subject to a fixed 100-day timeline, creating a narrow window for preparation. A final JCA report is prepared and formally endorsed within 30 days of the European Commission’s marketing authorisation decision.
This structure reinforces a key reality for developers. JCAs do not replace national HTA, but they fundamentally shape the clinical narrative on which national decisions are built.
PICO scoping sits at the heart of the JCA process. Assessors propose PICOs for consultation; stakeholders (e.g., clinical experts) provide input; and a consolidated set of PICOs determines the evidence requirements for the JCA dossier.
In practice, PICO volumes can still be substantial, particularly in oncology, where variation in comparators, biomarkers and treatment lines may define multiple subpopulations. While Regulation (EU) 2021/2282 aims to consolidate PICOs to the lowest feasible number, developers should plan for complexity.
This places a premium on evidence flexibility. Programmes that rely on a single pivotal comparison may not address all PICOs. Anticipating the need for indirect treatment comparisons and predefined subgroup analyses can help ensure timely and efficient JCA submissions.
Once scoping is finalised, developers have as little as 100 days to prepare and submit the JCA dossier (60 days in accelerated procedures and certain variations). This underscores the importance of having key analyses and methodologies planned in advance.
In parallel, national pre-positioning activities and EMA interactions continue, leading to regulatory, JCA, and national HTA workstreams increasingly overlapping. Strong cross-functional governance and early decision-making are critical to maintaining consistency across submissions and avoiding conflicting narratives.
JCAs are inherently comparative. Few development programmes will have head-to-head data against all relevant comparators. As a result, robust indirect treatment comparisons and network meta-analyses are likely to be central to many dossiers.
Where available, real-world evidence can support external controls, contextualise treatment pathways or address evidence gaps. Regardless of the approach, transparency, bias mitigation and sensitivity analyses are essential to maintain credibility at both EU and national levels.
The most resilient strategies are built around a single evidence system that can support EMA benefit-risk, JCA clinical effectiveness, and national HTA value assessments, rather than treating these as separate projects.
Alignment is best achieved early. Joint Scientific Consultations, ideally coordinated with EMA scientific advice, allow developers to discuss comparators, endpoints and analysis with both the CHMP and EU HTA bodies before pivotal protocols are finalised.
Pre-planning indirect comparison strategies, maintaining live systematic literature reviews, and preparing modular analysis code enable teams to respond quickly once final PICOs are confirmed. This reduces late-stage pressure and helps ensure methodological consistency across markets.
Although the JCA aims to standardise clinical assessment, national HTA bodies will continue to apply local context, economic evaluation and health system considerations. Variation in interpretation and implementation is likely, particularly in the early years.
Developers should therefore plan national value narratives that build on the JCA but remain tailored to country-specific decision frameworks. Preparing national adaptation kits in parallel with the JCA can significantly shorten timelines from EU endorsement to local reimbursement decisions.
The EU has also established the legal framework for joint clinical assessment of medical devices and in vitro diagnostics. While this article focuses on medicines, organisations with mixed portfolios should monitor evolving guidance and timelines as the HTA architecture expands beyond pharmaceuticals.
Regulatory and access success under the EU HTA framework will depend on early, integrated planning. In practice, this means:
DLRC’s Regulatory Aligned HTA and Market Access Support bridges the traditional gap between regulatory approval and national access. Our integrated approach is built around a single evidence system that can travel from EMA to JCA to national HTA without losing coherence or credibility. We support sponsors through:
The DLRC team led the first JSC procedure following the implementation of HTAR in the EU. To find out more about how we did this, read our project spotlight here.
The EU HTA Regulation and the introduction of Joint Clinical Assessments represent a significant shift in how clinical evidence is evaluated for market access across Europe. By centralising clinical assessment at the EU level while preserving national decision-making on pricing and reimbursement, the framework creates both opportunities and complexity for developers.
Success under the new regime depends on early, integrated evidence planning. PICO-driven scoping, compressed timelines and variable national implementation mean that late alignment is no longer viable. Organisations that invest early in cross-functional coordination, methodological readiness and modular evidence systems will be best positioned to navigate JCAs efficiently and support downstream reimbursement outcomes.
DLRC’s Regulatory Aligned HTA and Market Access Support is designed to help sponsors respond to this new environment with confidence. By aligning regulatory affairs and HTA strategy from the outset, we support evidence generation that meets regulatory standards while addressing payer expectations across Europe.
As the JCA framework continues to evolve, early planning and an integrated strategy will be critical. DLRC is ready to support developers at every stage, helping translate regulatory approval into sustainable market access and timely patient benefit across EU Member States.
This article reflects DLRC’s interpretation of the EU HTA framework and publicly available guidance.

Published Mar 20, 2026

Published Feb 26, 2026

Published Feb 26, 2026

Published Jan 07, 2026

Published Dec 02, 2025

Published Nov 27, 2025

Published Nov 25, 2025

Published Nov 24, 2025

Published Nov 24, 2025