EU HTA and JCA: Redefining Market Access Strategy in Europe
Published Feb 26, 2026
Published 29th July 2025

At its 180th session in November 2024, the European Pharmacopoeia Commission (EPC) made a landmark decision to completely remove the Rabbit Pyrogen Test (RPT) from all European Pharmacopoeia (Ph. Eur.) monographs. This change in legal requirements for the safety testing of medicinal products, published in Supplement 11.8, became official on 1 July 2025 and must be implemented by January 2026.
Pyrogens are fever-inducing substances that can cause serious side effects when administered to patients with a contaminated medicinal product or implanted medical device. The most common cause of pyrogenic reactions are bacterial endotoxins formed from the breakdown of gram-negative bacteria such as E.coli. Medicines that have used biological or fermentation methods in their production, have been exposed to microbial contamination during production, or where contaminated raw materials have been introduced, are at particular risk. These toxins can remain even after the product has been sterilised and pose a significant risk in injectable medicines.
The RPT has been a standard method for detecting pyrogens in parenteral (injectable) medicines since the 1970s. The test involves injecting a sample into rabbits to detect evidence of a temperature rise. While historically effective, the method has long been criticised for its ethical implications and reliance on animal testing. Its removal marks a significant advancement in both animal welfare and scientific innovation, aligning with the 3Rs principle (Replace, Reduce, Refine) and the growing adoption of non-animal models.
Marketing authorisation holders must therefore remove the RPT from their medicinal product dossiers. But what will replace it?
Researchers widely use the Bacterial Endotoxins Test (BET), a non-animal method, as an alternative screen for endotoxin pyrogens. However, companies should be aware that it does not detect pyrogens from other sources (non-endotoxin pyrogens). These contaminants may arise from gram-positive bacteria, fungi, viruses, and other particles. This is why experts must conduct a careful risk analysis and, depending on the outcome, may need to apply additional methods like the monocyte activation test (MAT). In this article, we outline the key steps that marketing authorisation holders need to take to ensure compliance and maintain product safety.
Scientists and regulators widely celebrate the move away from the RPT as a significant step forward in harmonising scientific rigour with ethical responsibility. It is estimated that 400,000 rabbits are used globally each year for pyrogen testing. The elimination of the RPT from the Ph. Eur. is expected to drastically reduce this number, setting a precedent for other regulatory bodies worldwide.
It is part of a longstanding effort by the EPC to Replace, Reduce and Refine (3Rs) the use of animals for test purposes. Animal tests were once seen as an essential part of product safety testing due to their assumed broad ability to detect unidentified contaminants. Advances in analytical techniques and manufacturing practices now reduce the need for these tests. The 2024 decision by the EPC also saw the removal of the final references to chapters on Histamine and Depressor substances, both of which also required the use of animals. This means the end of general animal safety testing in the Ph. Eur.
With the RPT removed from the Ph. Eur., marketing authorisation holders (MAHs) must now remove this test from their marketing authorisation dossiers and evaluate the need for alternative validated in vitro methods. As highlighted above, you should only use BET testing alone if you can rule out the presence of non-endotoxin pyrogenic substances. Where non-endotoxin pyrogens may be present, the Monocyte Activation Test (MAT), described in Ph. Eur. chapter 2.6.30, must be used.
The monocyte activation test uses human immune cells (monocytes), that respond to the presence of endotoxins or non-endotoxin pyrogens by secreting pro-inflammatory cytokines linked to the fever reaction. The released cytokines can be measured directly using standard techniques.
Therefore, MAHs should perform a risk analysis and assess the potential presence of pyrogens in their medicinal products. As part of the risk analysis, the team may perform an MAT test if necessary. Based on the outcome, they may decide to introduce routine MAT testing or to use the BET as the sole pyrogenicity test. The new Ph. Eur. general chapter 5.1.13 “Pyrogenicity” provides guidance on selecting appropriate methods based on a risk assessment.
The decision to eliminate the RPT reflects:
In summary, to comply with the new Ph. Eur. requirements, MAHs must take the following steps:
Pharmacopoeias have already validated the general test methods, so full re-validation is not necessary. But MAHs should perform product specific method verification for the selected in vitro test to demonstrate its suitability. Any non-pharmacopoeial methods need full validation.
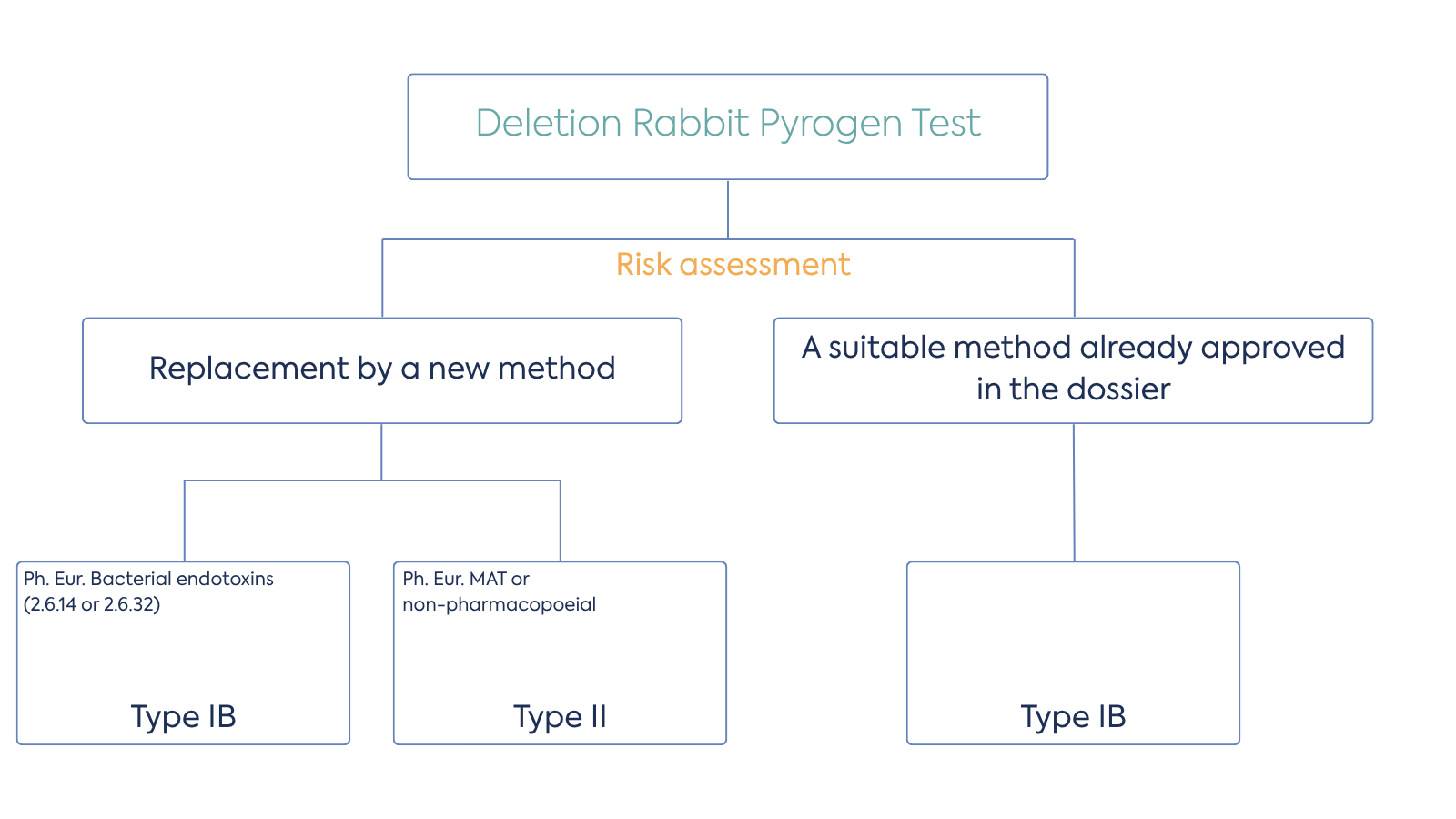
Figure 1. Decision tree for the deletion of the Rabbit Pyrogen Test from the EMA.
Some examples are provided below, but the list is not exhaustive:
Other changes include:
Supplement 11.8 of the Ph. Eur. has published the revised texts, which include the deletion of RPT references and the introduction of a new general chapter on Pyrogenicity (Chapter 5.1.13). These changes became official on 1 July 2025. MAHs must implement the new methods by 1 January 2026, as Ph. Eur. 11.8 comes into force.
The removal of the Rabbit Pyrogen Test from the European Pharmacopoeia is a progressive step toward modern, ethical, and scientifically robust pharmaceutical testing. Marketing authorisation holders must act promptly to ensure compliance by 1 January 2026, aligning their dossiers with the new alternative in vitro test method.
The changing requirements for nonclinical development increasingly use non-animal methods, reflecting the move to Replace, Reduce, and Refine animal use. As highlighted in our recent article on Innovating Non-Clinical Testing: NAMs and ESG Impact, these advancements not only support ethical testing practices but also align with broader Environmental, Social, and Governance (ESG) goals. The phasing out of batch-by-batch pyrogen testing reflects a welcome general trend.
DLRC’s regulatory experts have been closely monitoring the development of these regulations and are ready to assist marketing authorisation holders in navigating this new landscape. Our team can help optimise the use of non-animal testing methods while ensuring full compliance with all requirements. Contact us today to discuss how we can support you in this evolving regulatory environment by emailing hello@dlrcgroup.com

Published Feb 26, 2026

Published Jan 07, 2026

Published Dec 02, 2025

Published Nov 27, 2025

Published Nov 25, 2025

Published Nov 24, 2025

Published Nov 24, 2025

Published Nov 24, 2025

Published Nov 14, 2025